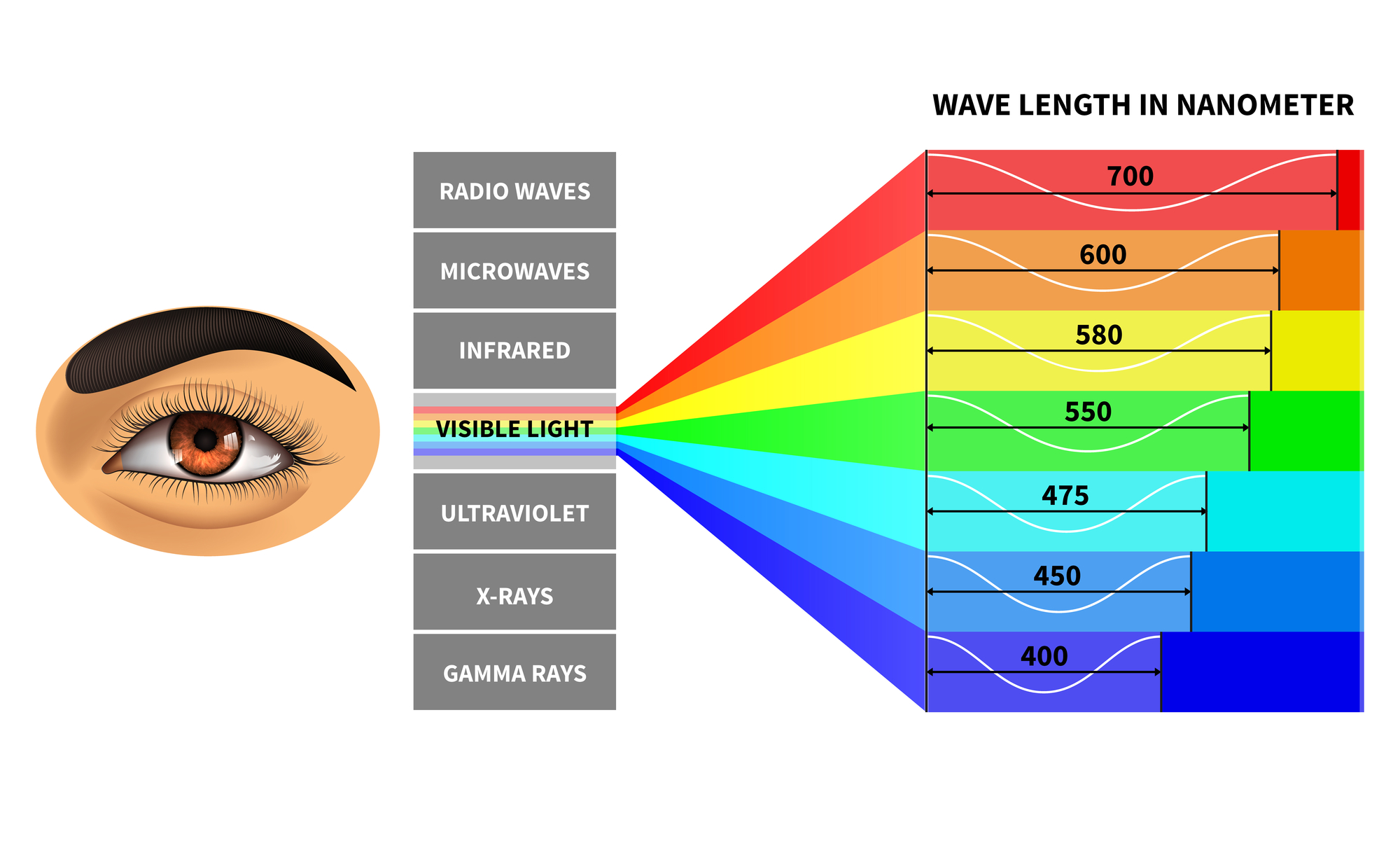
UV Radiation in Earth's AtmosphereĮarth's atmosphere prevents most UV radiation from space from reaching the ground. UV-C is sometimes called "Short Wave" UV or "germicidal" UV the latter because it is sometimes used to sterilize laboratory equipment or to purify water by killing microbes. These short-wavelength UV photons have high energies, and are very damaging to living creatures. The third region of the UV spectrum, UV-C, includes radiation with wavelengths between 100 and 280 nm. UV-B radiation is the main cause of sunburn the SPF factor listed on sunscreens refers to their ability to reduce the effects of UV-B. UV-B waves, with wavelengths between 280 and 320 nm, carry more energy than UV-A waves. Almost all of the ultraviolet radiation that makes it through our atmosphere to Earth's surface is UV-A. It is the closest UV radiation to visible light. UV-A, which is also called "blacklight" or "Long Wave" UV, spans wavelengths between 320 and 400 nm. You have probably seen UV-A and UV-B mentioned on the labels for sunglasses or sunscreen. They speak of the UV-A, UV-B, and UV-C regions of the UV spectrum. When discussing the impact of UV radiation on the environment and human health, scientists subdivide the ultraviolet spectrum in a different way. That is good news for us Earthlings, since our atmosphere shields us from the most dangerous, highest energy portions of the UV spectrum that reach our planet from the Sun and other sources in space. Normal air is largely opaque to UV with wavelengths shorter than 200 nm oxygen absorbs "light" in that part of the UV spectrum. Extreme UV radiation spans the 10 to 30 nm wavelength range. Extreme UV radiation has the shortest wavelength range and highest energies of the regions of the ultraviolet spectrum, and lies on the border between UV and X-ray radiation. The higher energy, shorter wavelength far UV region spans wavelengths between 91 and 200 nm. The near UV region lies closest to visible light, and includes wavelengths between 200 and 400 nm. These divisions are comparable to the partitions between different colors, and hence different wavelengths, of visible light. Scientists subdivide the ultraviolet spectrum into regions named near UV, far UV, and extreme UV. The UV portion of the spectrum has different regions, like the different colors of visible light, that correspond to specific wavelengths of UV radiation. Red light has a wavelength near 650 nm, while the wavelength of blue light is around 440 nm. When we talk about visible light, we refer to the different wavelengths of light across the visible spectrum by the names of colors. Ultraviolet radiation oscillates at rates between about 800 terahertz (THz or 10 12 hertz) and 30,000 THz. The wavelength of violet light is around 400 nanometers (or 4,000 Å). UV "light" spans a range of wavelengths between about 10 and 400 nanometers. Ultraviolet radiation lies between visible light and X-rays along the electromagnetic spectrum. Purple and violet light have shorter wavelengths than other colors of light, and ultraviolet has even shorter waves than violet does so ultraviolet is sort of "purpler-than-purple" light or "beyond violet" light. UV light has a shorter wavelength than visible light. Ultraviolet (UV) "light" is a type of electromagnetic radiation. Each of these energy packets is known as a quantum (plural: quanta).Blacklights such as this emit invisible ultraviolet (UV) "light" as well as the purple light that you can see. The reality, which Planck discovered, is that energy is not continuous but quantized-meaning that it can only be transferred in individual “packets” (or particles) of the size h ν h\nu h ν h, \nu. This was a shocking discovery, because it challenged the idea that energy was continuous, and could be transferred in any amount. 6 2 6 × 1 0 − 3 4 J ⋅ s 6, point, 626, times, 10, start superscript, minus, 34, end superscript, start text, space, J, end text, dot, start text, s, end text, and ν \nu ν \nu is the frequency of the light absorbed or emitted. Planck observed that matter actually absorbed or emitted energy only in whole-number multiples of the value h ν h\nu h ν h, \nu, where h h h h is Planck’s constant, 6.626 × 1 0 − 34 J ⋅ s 6.626\times10^ 6. Planck found that the electromagnetic radiation emitted by blackbodies could not be explained by classical physics, which postulated that matter could absorb or emit any quantity of electromagnetic radiation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
